Gesynta Pharma’s drug candidates utilize a unique mechanism of action to reduce inflammation and pain. By targeting mPGES-1, a key enzyme present at sites of inflammation, our drug candidates attenuate the inflammatory process with substantially higher precision than currently available treatments. Read more about our drug candidates and how they work below.

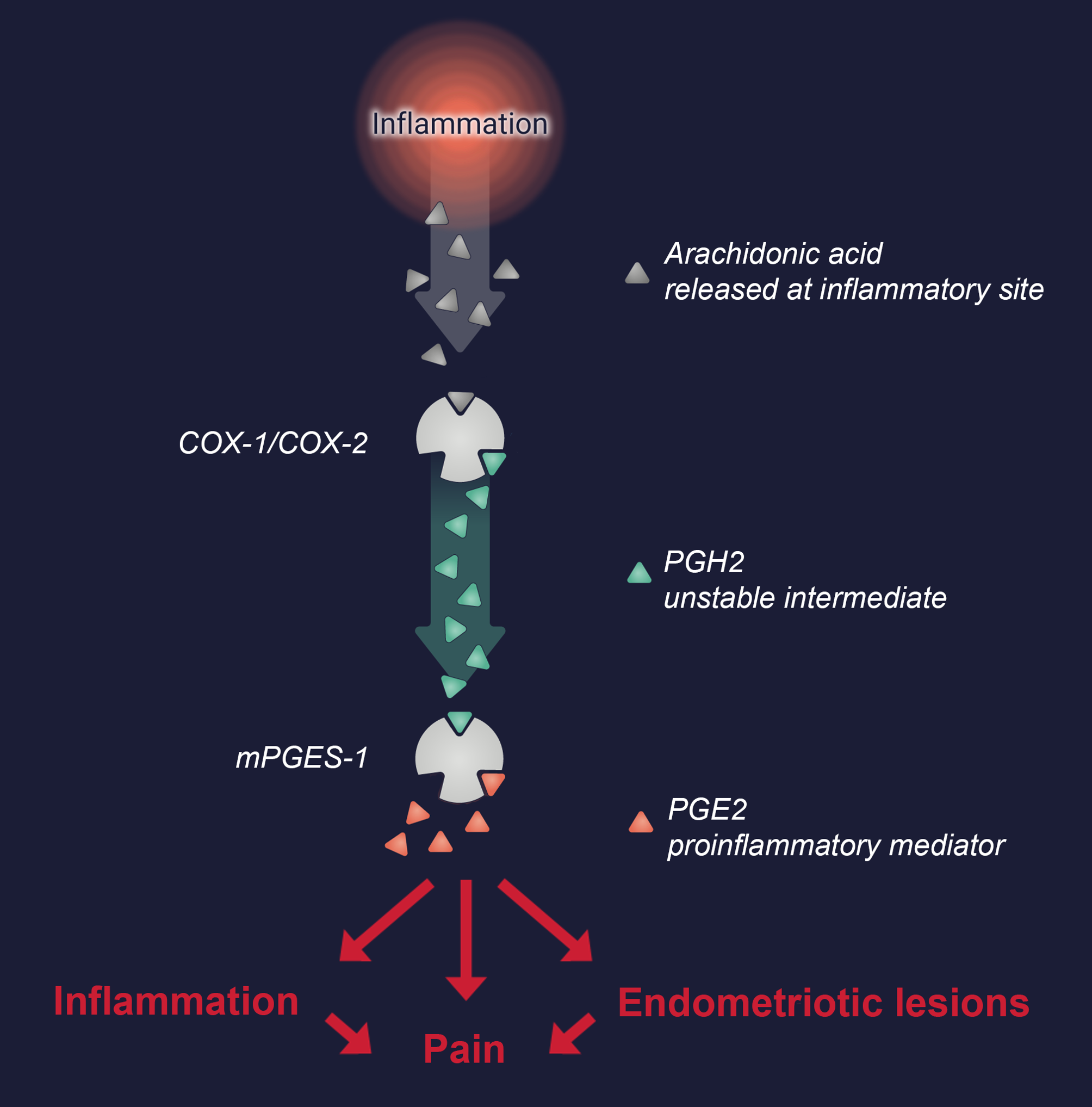
Gesynta Pharma’s drug candidates have a unique ability to selectively inhibit microsomal prostaglandin E synthase (mPGES-1), the enzyme that promotes production of the proinflammatory mediator prostaglandin E2 (PGE2). By inhibiting mPGES-1, the levels of PGE2 decrease, resulting in anti-inflammatory and pain-relieving effects.
Read more about vipoglanstat, in Phase 2 för the treatment of endometriosis
Read more about GS-073 for the treatment of chronic inflammatory pain