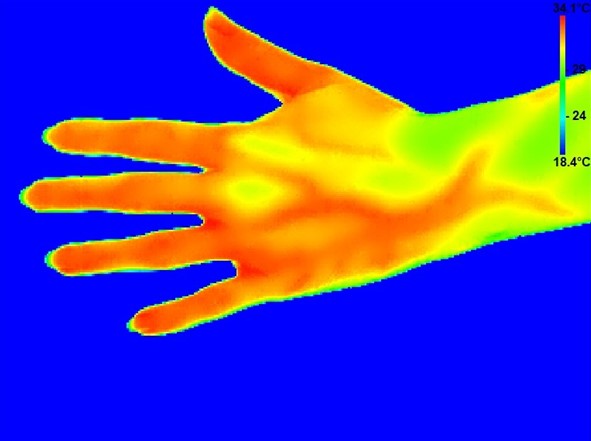
We are pleased to announce the publication of our clinical Phase II study in the prestigious journal Rheumatology. The study, conducted from 2020 to 2022, aimed to determine the safety and efficacy of vipoglanstat (GS-248) versus placebo on Raynaud's phenomenon in subjects with the chronic inflammatory disease systemic sclerosis. Over the four-week treatment course, vipoglanstat demonstrated excellent tolerability, a favorable safety profile and complete inhibition of the target enzyme mPGES-1. In fact, once daily administration of 120 mg vipoglanstat resulted in systemic exposure levels well exceeding those needed to achieve full inhibition of mPGES-1, while the adverse event profile remained similar to that of placebo. However, no significant impact on Raynaud's phenomenon or peripheral blood flow was observed.
In parallel, we have generated excellent data in an advanced disease model of endometriosis, confirming previously published support for the involvement of mPGES-1 and PGE2 in endometriosis pathophysiology. Thus, we are now in the final stages of planning for a Phase II study in endometriosis.
Finally, we want to express our deepest gratitude to the investigator teams and the study participants who generously participated in this well-conducted study.
https://academic.oup.com/rheumatology/advance-article/doi/10.1093/rheumatology/keae049/7593822
Image for illustrative purposes only.